About This Solution Guide
Welcome to the complete solution guide for Madhyamik 2017 Physical Science question paper! This resource is designed to help Class 10 students understand concepts thoroughly, not just memorize answers.
Each solution includes detailed explanations to ensure you grasp the underlying principles. Whether you're preparing for exams or revising concepts, this guide will be your perfect companion.
Key Features
- Complete question paper coverage
- Step-by-step solutions
- Concept explanations
- Interactive question navigation
- Mobile-friendly design
📝 Exam Preparation Tip
Focus on understanding concepts rather than memorizing answers. This approach will help you tackle similar questions even if they're framed differently in your exam.
GROUP-A: MCQ Questions
(a) methane
(b) water vapour
(c) carbon dioxide
(d) oxygen
(a) Nm\(^2\)
(b) Nm\(^3\)
(c) Nm
(d) N
(a) 8
(b) 16
(c) 32
(d) 64
(a) temperature
(b) length
(c) nature of the material
(d) area of the cross-section
a. f=2r
b. f=r/2
c. f=r/3
d. f=(2/3) r
(a) red
(b) yellow
(c) violet
(d) green
(a) coulomb-second
(b) volt-ohm\(^{-1}\)
(c) volt-ohm
(d) volt\(^2\)-ohm
(a) conductor
(b) semiconductor
(c) superconductor
(d) insulator
(a) \( \alpha > \beta > \gamma \)
(b) \( \alpha > \gamma > \beta \)
(c) \( \gamma > \beta > \alpha \)
(d) \( \beta > \alpha > \gamma \)
(a) K
(b) H
(c) Li
(d) Na
(a) hydrogen chloride
(b) sodium chloride
(c) lithium hydride
(d) calcium oxide
(a) CH\(_3\)COOH
(b) NaOH
(c) H\(_2\)SO\(_4\)
(d) NaCl
(a) conc. H\(_2\)SO\(_4\)
(b) P\(_2\)O\(_5\)
(c) CaO
(d) CaCl\(_x\)
(a) bauxite
(b) haematite
(c) malachite
(d) chalcopyrites
a. -OH
b. -CHO
c. -COOH
d. =CO
GROUP-B: Short Answer Questions
| Left Column | Right Column |
| 2.11.1 A transition element | (a) Aluminium |
| 2.11.2 A transuranium element | (b) Nickel |
| 2.11.3 Reduces Fe\(_2\)O\(_3\) at high temperature in the thermit process | (c) Tin |
| 2.11.4 Present in the alloy bell metal | (d) Plutonium |
| Left Column | Right Column |
| 2.11.1 A transition element | (b) Nickel |
| 2.11.2 A transuranium element | (d) Plutonium |
| 2.11.3 Reduces Fe\(_2\)O\(_3\) at high temperature in the thermit process | (a) Aluminium |
| 2.11.4 Present in the alloy bell metal | (c) Tin |
Explanation:
• Nickel is a transition element (d-block element)
• Plutonium is a transuranium element (atomic number > 92)
• Aluminium reduces iron oxide in thermit process: \( 2\text{Al} + \text{Fe}_2\text{O}_3 \rightarrow 2\text{Fe} + \text{Al}_2\text{O}_3 \)
• Bell metal is an alloy of copper and tin
Fluorine atom (atomic number 9) has electronic configuration: 2,7
It needs 1 electron to complete its octet
In F\(_2\) molecule, two fluorine atoms share one pair of electrons
Lewis structure: :F ─ F:
Each fluorine atom has 3 lone pairs (6 electrons) and 1 shared pair (2 electrons)
• Cu\(^{2+}\) ions (cations) migrate towards cathode
• At cathode: Cu\(^{2+}\) + 2e\(^-\) → Cu (copper deposition)
• SO\(_4^{2-}\) ions (anions) migrate towards anode
• At anode: Cu → Cu\(^{2+}\) + 2e\(^-\) (copper dissolution)
Pb(NO\(_3\))\(_2\) + H\(_2\)S → PbS ↓ + 2HNO\(_3\)
Black precipitate of lead sulfide (PbS) is formed.
Orange color of dichromate (Cr\(_2\)O\(_7^{2-}\)) changes to green color of chromium(III) ions (Cr\(^{3+}\)).
K\(_2\)Cr\(_2\)O\(_7\) + 4H\(_2\)SO\(_4\) + 3H\(_2\)S → K\(_2\)SO\(_4\) + Cr\(_2\)(SO\(_4\))\(_3\) + 7H\(_2\)O + 3S
2NH\(_3\) + CO\(_2\) → NH\(_2\)CONH\(_2\) + H\(_2\)O
This reaction occurs under high pressure (150-250 atm) and temperature (180-200°C).
• Water pipes and plumbing fittings
• Electrical cable insulation
• Flooring materials
• Window frames
• Medical devices
• Packaging materials
Both have same molecular formula C\(_2\)H\(_6\)O but different functional groups (alcohol vs ether).
GROUP-C: Short Answer Questions
Using Boyle's law: \( P_1V_1 = P_2V_2 \)
At STP, P₁ = 76 cmHg, V₁ = 52 m³
P₂ = 104 cmHg
\( V_2 = \frac{P_1V_1}{P_2} = \frac{76 \times 52}{104} = 38 \) m³
Using ideal gas equation: \( PV = nRT \)
Molar mass of H₂ = 2 g/mol
Number of moles, \( n = \frac{8}{2} = 4 \) moles
\( V = \frac{nRT}{P} = \frac{4 \times 0.082 \times 300}{4} = 24.6 \) litres
1. Higher efficiency: CFLs convert about 20-25% of electrical energy into visible light, while incandescent bulbs convert only about 5%.
2. Longer lifespan: CFLs last 8-15 times longer than incandescent bulbs (approx. 8000 hours vs 1000 hours).
Additional advantages: Lower heat generation, energy saving, cost-effective in long run.
Melting point: Sodium chloride has a high melting point (801°C) due to strong ionic bonds. Naphthalene has a lower melting point (80°C) due to weaker intermolecular forces.
Solubility in water: Sodium chloride is highly soluble in water due to ion-dipole interactions. Naphthalene is insoluble in water as it is non-polar and cannot form hydrogen bonds with water.
Second Part Kipp's apparatus is used to prepare gases by the action of a liquid on a solid. Hydrogen sulfide can be prepared by the action of dilute HCl on iron sulfide:
\( \text{FeS} + 2\text{HCl} \rightarrow \text{FeCl}_2 + \text{H}_2\text{S} \uparrow \)
Note: Other gases that can be prepared in Kipp's apparatus: CO₂ (using marble chips and HCl), H₂ (using zinc and HCl).
\( 2\text{ZnO} + \text{C} \rightarrow 2\text{Zn} + \text{CO}_2 \)
\( \text{Zn} + \text{CuSO}_4 \rightarrow \text{ZnSO}_4 + \text{Cu} \)
According to electronic theory:
- Zinc loses 2 electrons: \( \text{Zn} \rightarrow \text{Zn}^{2+} + 2e^- \) (Oxidation)
- Copper gains 2 electrons: \( \text{Cu}^{2+} + 2e^- \rightarrow \text{Cu} \) (Reduction)
This is a redox reaction where zinc is oxidized and copper is reduced.
H₂C=CH₂
The presence of a carbon-carbon double bond makes it unsaturated. Unsaturated hydrocarbons have double or triple bonds and can undergo addition reactions.
GROUP-D: Long Answer Questions
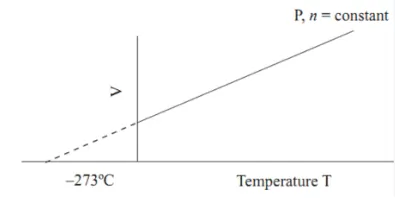
Charles' Law: At constant pressure, the volume of a fixed mass of gas is directly proportional to its absolute temperature.
Mathematically: \( V \propto T \) or \( \frac{V}{T} = \text{constant} \)
Graphical representation:
Reaction: \( (\text{NH}_4)_2\text{SO}_4 + 2\text{NaOH} \rightarrow \text{Na}_2\text{SO}_4 + 2\text{NH}_3 + 2\text{H}_2\text{O} \)
Molar mass of NH₃ = 14 + 1×3 = 17 g/mol
Molar mass of (NH₄)₂SO₄ = (14 + 1×4)×2 + 32 + 4×16 = 132 g/mol
From equation: 2 moles NH₃ (34g) from 1 mole (NH₄)₂SO₄ (132g)
For 6.8g NH₃, required (NH₄)₂SO₄ = \( \frac{132}{34} \times 6.8 = 26.4 \) g
Reaction: \( \text{CaCO}_3 + 2\text{HCl} \rightarrow \text{CaCl}_2 + \text{CO}_2 + \text{H}_2\text{O} \)
Molar mass HCl = (1 + 35.5) g/mol = 36.5 g/mol
From stoichiometry: 1 mole CaCO₃ \((=40+12+3 \times 16 =100g)\) requires 2 moles HCl \((2\times 36.5 = 73g)\)
For 50g CaCO₃, required HCl = \( \frac{73}{100} \times 50 = 36.5 \) g
But we need to produce specific amounts:
From 55.5g CaCl₂ (0.5 mole), we need 1 mole HCl = 36.5g
From 22.2g CO₂ (0.5 mole), we need 1 mole HCl = 36.5g
From 9.0g H₂O (0.5 mole), we need 1 mole HCl = 36.5g
Total HCl required = 36.5g
Moles of HCl = \( \frac{36.5}{36.5} = 1 \) mole
1. Both follow similar mathematical equations (Fourier's law for heat, Ohm's law for electricity)
2. Both depend on material properties (thermal conductivity and electrical conductivity)
Non-metal with high thermal conductivity: Diamond has the highest thermal conductivity among non-metals (about 5 times that of copper).
Second Law of Refraction (Snell's Law): The ratio of the sine of the angle of incidence to the sine of the angle of refraction is constant for a given pair of media.
Mathematical formula: \( \frac{\sin i}{\sin r} = \text{constant} = \mu \)
Where \( \mu \) is the refractive index of the second medium with respect to the first medium.
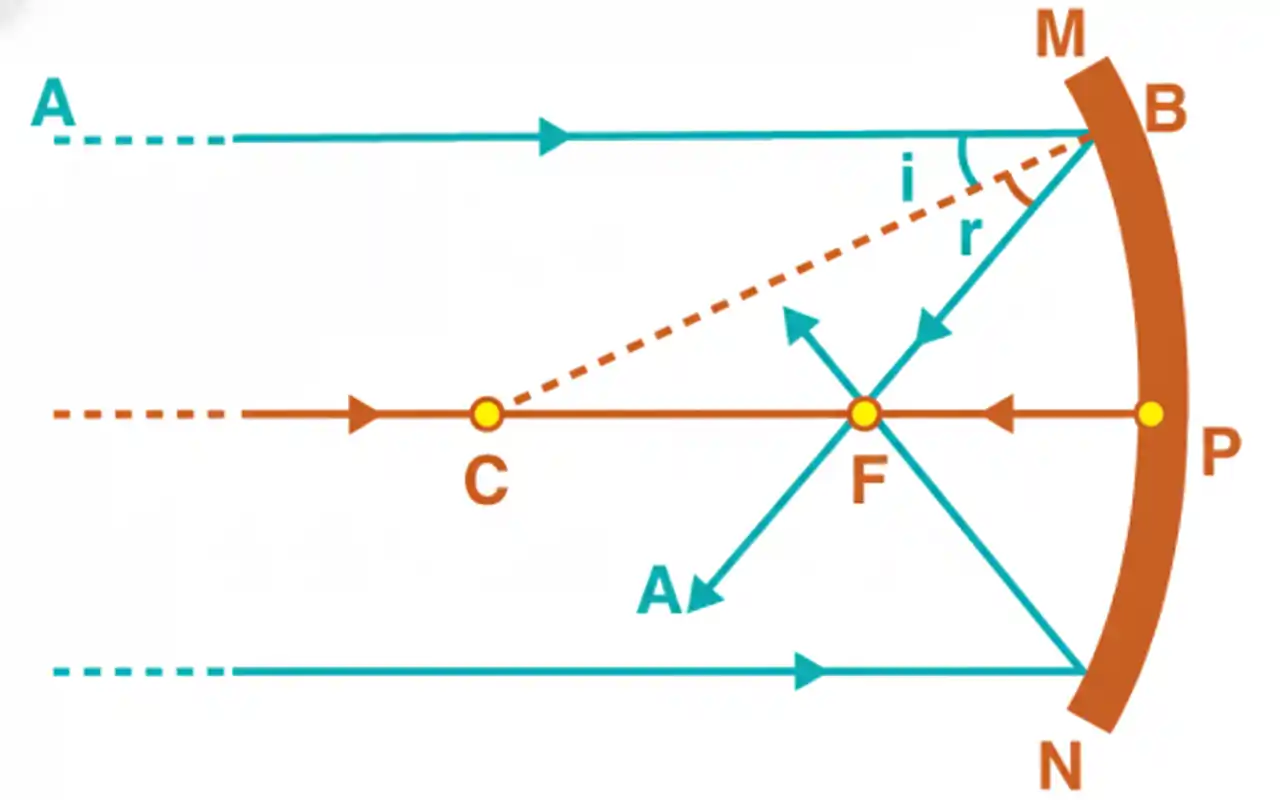
A light ray AB strikes a spherical mirror at point B while being parallel to the principal axis. CB serves as the normal line to the surface at point B. The radius of curvature is denoted as CP = CB = R. After the light reflects off the mirror, it converges at the focus of the concave mirror F, following the law of reflection, which states that the angle of incidence equals the angle of reflection (i = r).
From the geometry of the figure,
∠BCP = θ = i (As ∠BCP and ∠ABC are alternate angles)
In ΔCBF, ∠CBF = θ = r
∴ BF = FC (because i = r)
If the aperture of the mirror is small, B lies close to P, and therefore BF = PF
=> FC = BF = PF
=> PC = PF + FC = PF + PF
=> R = 2 PF = 2f
=> f = R/2 (Hence Proved)
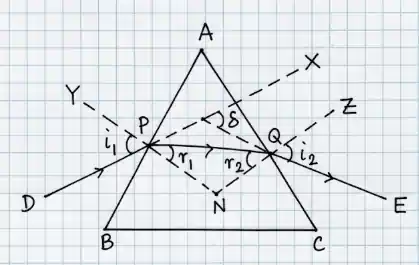
In quadrilateral APNQ: \( \angle A + \angle PNQ = 180^\circ \) ...(1)
In triangle PNQ: \( r_1 + r_2 + \angle PNQ = 180^\circ \) ...(2)
From (1) and (2): \( A = r_1 + r_2 \)
Angle of deviation: \( \delta = (i_1 - r_1) + (i_2 - r_2) \)
\( \delta = i_1 + i_2 - (r_1 + r_2) \)
\( \delta = i_1 + i_2 - A \)
Faraday's Second Law: The magnitude of the induced emf is directly proportional to the rate of change of magnetic flux linked with the closed coil.
Mathematically: \( \epsilon = -N \frac{d\Phi}{dt} \)
Where \( \epsilon \) is induced emf, N is number of turns, and \( \frac{d\Phi}{dt} \) is rate of change of magnetic flux.
Alternating Current (AC): An electric current that periodically reverses direction and changes its magnitude continuously with time, in contrast to direct current (DC) which flows only in one direction.
Energy consumed = Power × Time = 0.11 kW × 10 h = 1.1 kWh (BOT unit)
When length becomes double (2l), volume remains constant, so area becomes half (A/2)
New resistance, \( R' = \rho \frac{2l}{A/2} = 4 \times \rho \frac{l}{A} = 4 \times 6 = 24 \) ohm
Source of energy in nuclear fusion: In nuclear fusion, when light nuclei combine to form a heavier nucleus, the mass of the product nucleus is less than the sum of masses of the original nuclei. This mass defect is converted into energy according to Einstein's equation \( E = \Delta m c^2 \), where \( \Delta m \) is the mass defect and c is the speed of light.
Example: Atomic radius decreases across a period (from left to right) and increases down a group.
Non-periodic property: Radioactivity is not a periodic property.
Trend in Group 1: Electronegativity decreases from top to bottom in Group 1 (alkali metals). This is because atomic size increases down the group, reducing the nucleus's ability to attract bonding electrons.
Energy source: Electrical energy is responsible for the chemical reactions during electrolysis. It provides the necessary energy to drive non-spontaneous redox reactions.
Electrolysis of acidulated water (water with few drops of H₂SO₄):
At cathode (reduction): \( 2\text{H}^+ + 2e^- \rightarrow \text{H}_2 \uparrow \)
At anode (oxidation): \( 2\text{H}_2\text{O} \rightarrow \text{O}_2 \uparrow + 4\text{H}^+ + 4e^- \)
Overall reaction: \( 2\text{H}_2\text{O} \rightarrow 2\text{H}_2 \uparrow + \text{O}_2 \uparrow \)
\( 2\text{SO}_2 + \text{O}_2 \xrightarrow[400-450^\circ\text{C}]{\text{V}_2\text{O}_5} 2\text{SO}_3 \)
Conditions: Temperature 400-450°C, Pressure 1.5-1.7 atm, Vanadium pentoxide (V₂O₅) catalyst
Preparation of H₂SO₄ from SO₃:
SO₃ is dissolved in 98% H₂SO₄ to form oleum (H₂S₂O₇):
\( \text{SO}_3 + \text{H}_2\text{SO}_4 \rightarrow \text{H}_2\text{S}_2\text{O}_7 \)
Oleum is then diluted with water to get concentrated H₂SO₄:
\( \text{H}_2\text{S}_2\text{O}_7 + \text{H}_2\text{O} \rightarrow 2\text{H}_2\text{SO}_4 \)
\( \text{HC} \equiv \text{CH} + \text{H}_2 \xrightarrow[\text{Ni catalyst}]{200-300^\circ\text{C}} \text{H}_2\text{C} = \text{CH}_2 \) (ethene)
\( \text{H}_2\text{C} = \text{CH}_2 + \text{H}_2 \xrightarrow[\text{Ni catalyst}]{200-300^\circ\text{C}} \text{CH}_3 - \text{CH}_3 \) (ethane)
Use of LPG: As domestic fuel for cooking and heating.
\( \text{CH}_3\text{CH}_2\text{OH} \xrightarrow[\text{Conc. H}_2\text{SO}_4]{170^\circ\text{C}} \text{CH}_2 = \text{CH}_2 + \text{H}_2\text{O} \)
This is dehydration reaction (removal of water molecule).
Biodegradation of protein: Proteins are broken down by microorganisms through enzymatic hydrolysis into amino acids, which are further decomposed to simpler compounds like CO₂, H₂O, and NH₃.
GROUP-E: For External Candidates
Radioactive ray: Gamma rays are electromagnetic radiation, not particles. Alpha and beta rays consist of particles (helium nuclei and electrons respectively).
CNG source: Compressed Natural Gas (CNG) is obtained from natural gas fields. It is primarily methane (CH₄) compressed to less than 1% of its volume at standard atmospheric pressure.
1. Easy voltage transformation: AC voltage can be easily stepped up or down using transformers, enabling efficient long-distance transmission.
2. Lower transmission losses: At high voltages (used for transmission), AC has lower energy losses compared to DC.
Additional advantages: AC motors are simpler and cheaper, AC can be easily generated.
\( \text{FeCl}_3 + 3\text{NH}_4\text{OH} \rightarrow \text{Fe(OH)}_3 \downarrow + 3\text{NH}_4\text{Cl} \)
The reddish-brown precipitate is insoluble ferric hydroxide [Fe(OH)₃].
\( \text{CH}_4 + 2\text{O}_2 \rightarrow \text{CO}_2 + 2\text{H}_2\text{O} + \text{Heat} \)
This is an exothermic reaction used in natural gas as fuel.
Final Preparation Tips
Concept Clarity
Focus on understanding fundamental concepts rather than rote learning.
Time Management
Practice solving papers within the allotted time to improve speed.
Revision
Regular revision of important formulas and concepts is crucial.